Aisa Pharma is focused on developing a novel treatment for the pressing medical needs of patients with Systemic Sclerosis (or Scleroderma) who demonstrate Raynaud’s Phenomenon. Preliminary clinical data is encouraging and implies treatment effects of our drug on both Raynaud’s and aspects of the underlying disease and associated symptoms.
About AISA Pharma
A late-stage, clinical development company delivering new treatments for severe autoimmune and vascular diseases.
About AISA-021
AISA-021 is being developed to treat Systemic Sclerosis and Raynaud’s Phenomenon.
Our Team
The people at AISA Pharma have over 100 years of experience bringing new drugs to market.
Contact Us
Get in touch! AISA Pharma is located in Boston with offices in California and Australia. Investor information is available.
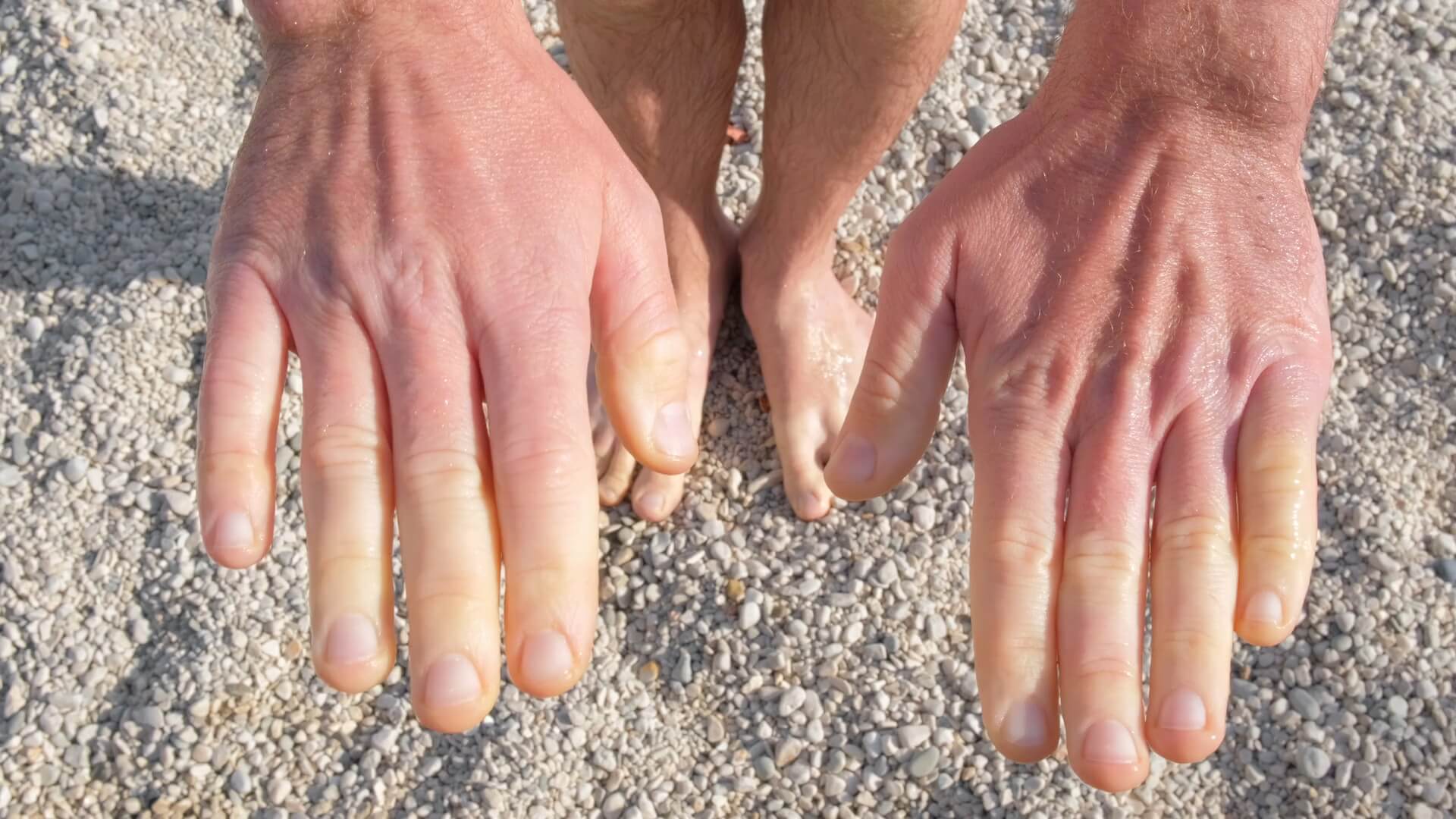
Raynaud syndrome, also known as Raynaud’s phenomenon, is a medical condition in which the spasm of small arteries causes episodes of reduced blood flow to end arterioles. Typically the disease occurs in the fingers, and less commonly, the toes and other extremities. The episodes classically result in the affected part turning white, blue, then red. Although usually self-limited, severe recurrent episodes can result in gangrene and amputation.
Aisa Pharma’s AISA-021 is a novel 4th generation dual channel calcium blocker being developed for the treatment of recurrent episodes of Raynaud’s phenomenon in patients with systemic sclerosis, the most severe form of Scleroderma.